Our Research
Building protein tools to decode the brain’s chemical language
The brain communicates through chemicals called neurotransmitters. These tiny messengers pass signals between neurons, helping control everything from movement and learning to mood and decision-making.
To truly understand how the brain works, we need to see when, where, and how these signals are released during behavior. This is especially important because many neurotransmitters, and the receptors that detect them, are involved in neurological and psychiatric disorders and are major targets for medicines.
Our work focuses on building protein-based tools that make these hidden chemical signals visible in living animals. By tracking neurotransmitter dynamics with high precision in space and time, we aim to uncover how brain chemistry, neural activity, and behavior are linked.
To better understand how neurons communicate, we need new molecular tools alongside advances in brain imaging. One of the most powerful examples is genetically encoded fluorescent sensors - proteins that light up when specific biological signals occur. Among these, GCaMP calcium sensors have become essential because they work well in living animals and can be combined with cutting-edge imaging methods.
Building on this foundation, our work and that of others has helped create the first genetically encoded sensors for dopamine, a neurotransmitter best known for its roles in reward, motivation, and learning. One example is dLight1, a highly sensitive dopamine sensor designed to make this chemical signal visible in real time.
These sensors are created by engineering a fluorescent protein into G protein-coupled receptors (GPCRs), the natural proteins cells use to detect neurotransmitters. This design allows the sensor to convert chemical binding into light, making it possible to watch neurotransmitter signaling unfold in living brains.
We engineer new optogenetic tools and approaches
to investigate neurotransmitter signaling at the system level
We are building the next generation of neurotechnology tools to track the molecules that enable brain cells to communicate in living animals. These tools will help us understand how neurotransmitters and their receptors influence behavior, brain function, and disease.
Our ultimate goal is to uncover the molecular basis of prevalent psychiatric disorders such as anxiety and depression. Since membrane receptors are among the most important targets in modern medicine, we also see these technologies as powerful tools for discovering and improving new drugs.
Ongoing projects
Our research combines state-of-the-art molecular biology, custom screening assays, ex vivo and in vivo brain imaging, automated behavior tracking, and mouse behavioral studies. We also actively build cross-disciplinary collaborations to share the tools and expertise we develop.
Our current projects focus on four main goals:
1. Building new genetically encoded sensors
We develop highly sensitive tools to detect neurotransmitters in cells, tissues, and living animals.
2. Creating new optogenetic and chemical biology tools
We design methods to precisely control signaling through the brain’s own receptors.
3. Using these tools to understand brain signaling
We study how neuromodulatory signals are organized across space and time in both healthy and diseased brains.
4. Developing new drug discovery platforms
We establish ultra-high-throughput screening systems to identify receptor-targeting molecules with potential for treating or preventing human disease.
Highlights from preprints and published work
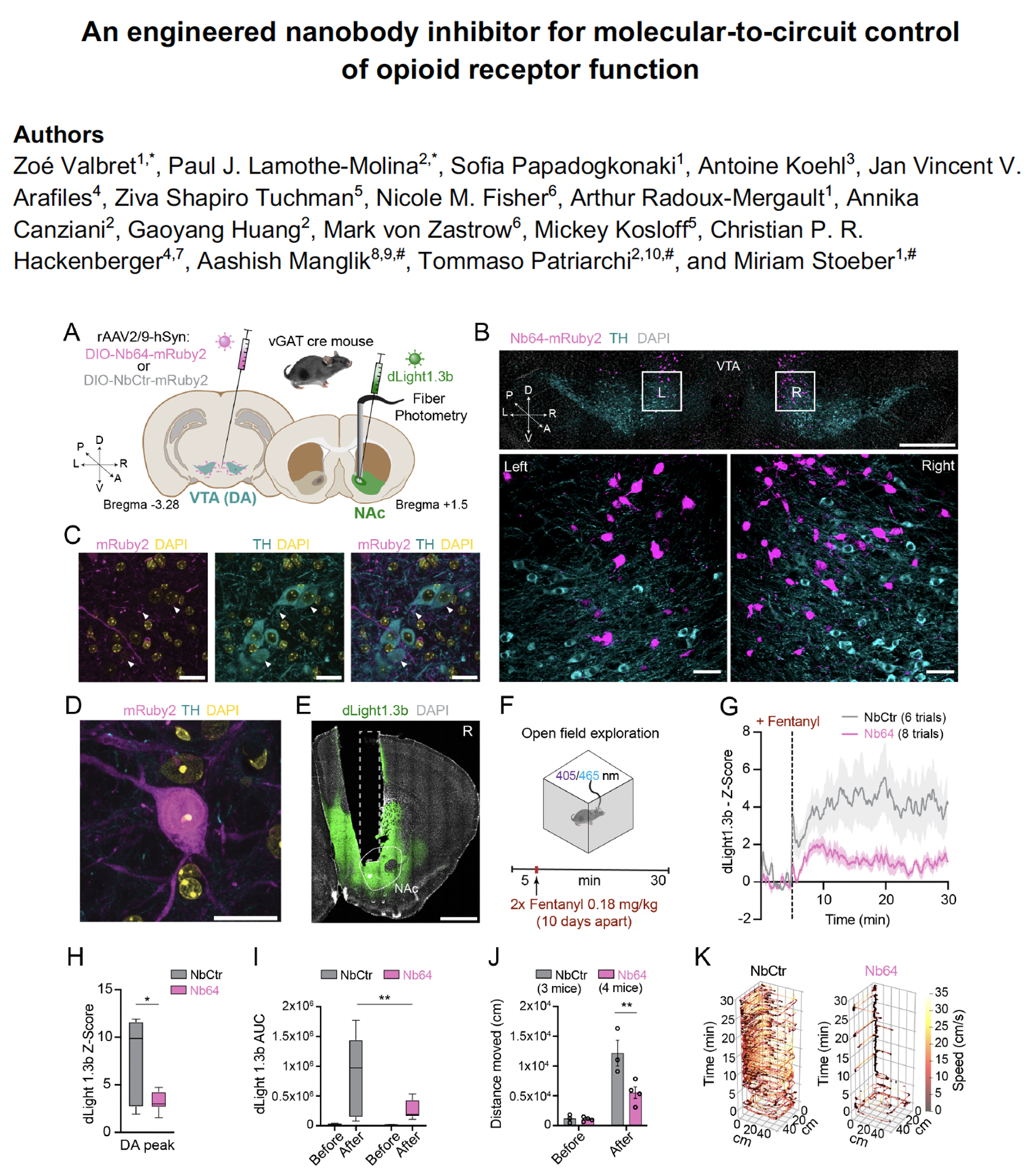
An engineered nanobody inhibitor for molecular-to-circuit control of opioid receptor function
We develop and apply genetically encoded tools to understand how neuromodulatory signals are represented across molecules, cells, and circuits.
In this study, we contributed the in vivo neuroscience arm by testing whether an intracellular nanobody called Nb64 could be used to control endogenous opioid receptor signaling in defined neuronal populations. We expressed Nb64 selectively in VTA GABAergic neurons and combined this manipulation with dLight1.3b fiber photometry in the nucleus accumbens, histological validation, and automated behavioral tracking during fentanyl exposure. This allowed us to link the molecular action of Nb64 to dopamine dynamics and behavior in awake mice.
Our experiments showed that cell-type-specific intracellular inhibition of μ-opioid receptor signaling attenuates fentanyl-evoked dopamine release and locomotor activation, demonstrating how engineered protein tools can connect receptor-level mechanisms to neuromodulatory circuit function in vivo.
Next-generation norepinephrine indicators for in vivo imaging
We developed nLightG2 and nLightR2, next-generation fluorescent sensors that make it easier to detect norepinephrine in the living brain. By improving both sensitivity and color flexibility, these tools allow norepinephrine release to be monitored more reliably across a wide range of experiments.
These sensors support dual-color imaging of norepinephrine together with neuronal or astrocytic activity, making it possible to study how this key neuromodulator shapes brain states and behavior. Using them, we tracked norepinephrine dynamics during sleep, fear learning, and hippocampal activity, and uncovered highly localized norepinephrine release events in the visual cortex of awake mice.
This work provides an improved multicolor toolkit for dissecting the fine spatiotemporal organization of norepinephrine signaling in the brain.
Watch norepinephrine (NE) release in real time in the
mouse visual cortex (layer 2/3) during spontaneous behavior.
These are in vivo head-fixed two-photon microscopy data
using our latest green NE sensor nLightG2.
Allosteric modulation of a dopamine sensor
In this project, we developed a new way to increase the sensitivity of the dopamine sensor dLight.
Our approach uses a highly specific molecule, originally developed by Eli Lilly, that acts as a positive allosteric modulator of the human dopamine D1 receptor.
We showed that applying this molecule to the system being studied—whether cultured dopaminergic neurons, brain slices, or living animals—can flexibly tune the sensitivity of dLight. This makes it possible to detect dopamine signals more effectively and, when needed, distinguish between tonic dopamine levels and phasic bursts of release.
A sensor for in vivo imaging of the opioid peptide nociceptin
In this project, we developed a new genetically encoded sensor to detect nociceptin, an opioid-related neuropeptide also known as orphanin FQ. This tool allows us to monitor nociceptin signaling in real time.
Using this sensor, we detected natural nociceptin release during acute stress and in behavioral experiments that measure an animal’s motivation to seek reward. By making this signaling pathway visible, NOPLight1 opens new ways to study how nociceptin interacts with neural activity to shape motivation, stress, and anxiety-related behavior.
Rapid and efficient sensor development by protein grafting
In this work, we used a modular grafting strategy to make sensor development faster, more scalable, and more efficient. By transferring key design elements across related receptor scaffolds, we were able to rapidly build and optimize new genetically encoded sensors for multiple signaling molecules.
Using this approach, we developed and validated new multicolor norepinephrine sensors, nLightG and nLightR, based on an alpha-1 adrenergic receptor scaffold. These sensors detect norepinephrine in living animals with high sensitivity, strong specificity, and fast temporal resolution. As part of the same effort, we also generated new sensors for acetylcholine, histamine, adenosine, and fractalkine, showing how this modular strategy can accelerate the expansion of the neurotechnology toolbox.
A sensor for GLP-1 and blockbuster weight-loss drugs
In this project, we developed GLPLight1, a new genetically encoded sensor based on the human GLP-1 receptor. This sensor makes it possible to measure receptor activation with high sensitivity and fast temporal resolution, while accurately capturing how strongly different drugs activate the receptor.
Using GLPLight1, we also established an all-optical assay to study Photo-GLP1, a newly developed light-activatable form of GLP-1. This allowed us to control GLP-1 receptor activation with light and monitor the response in real time.
Together, GLPLight1 and Photo-GLP1 create a powerful new optical toolkit for studying GLP-1 receptor signaling with precise control in both space and time.
Make it stand out
A synthetic light-activated orexin neuropeptide
We developed Photo-OXB, the first photocaged version of orexin-B, to enable light-controlled studies of orexin signaling. To make this possible, we created a new C-terminal photocaging strategy for peptides—an approach that could be extended to many other peptide signaling systems.
Using all-optical assays, we showed that Photo-OXB works seamlessly with biosensor imaging and can be activated with light to drive receptor signaling. We then applied this tool in brain slices, where light-triggered uncaging activated endogenous orexin receptors and changed the electrical state of neurons.
This work delivers both a new tool for probing the diverse roles of orexin signaling and a broadly useful method for developing and characterizing other light-activatable peptides.
A sensor for in vivo imaging of orexin neuropeptides
We developed OxLight1, a new genetically encoded sensor that makes it possible to detect orexin neuropeptides in real time. Built from the human type 2 orexin receptor, OxLight1 provides a highly sensitive and specific readout of orexin signaling in living systems.
OxLight1 responds to both orexin-A and orexin-B with nanomolar sensitivity, fast activation, and high specificity, making it an ideal tool for probing endogenous orexin signaling in living animals.
Find our plasmids and viruses on Addgene:
https://www.addgene.org/Tommaso_Patriarchi/